- Blog
- Dewalt 18v 20v battery adapter
- Diffraction of sound and light waves
- Expense tracker checklist printable
- Minecraft bedrock hack client 1-12
- Minecraft animated paintings texture pack
- Periodic table charge and molar mass
- The seven habits of highly effective people
- Ultimate chicken horse download 2019
- Simple expense tracker printable
- Skyrim console commands all alchemy recipes
- Camtasia 9 library assets free download
- Free download video effects for movie maker
- Cool text art copy and paste
So how good is our atomic radius? Standard tables give the atomic radius of strontium is in the range 192-220 pm. Since 3 3=27 and 4 3 = 64, you know that the cube root of 55 will be between 3 and 4, so the cube root should be a bit less than 4 × 10 –8.
These soft lewis acids prefer the company of soft lewis bases such as negatively charged (reduced) sulfur. To a chemist, this is referred to as 'soft (or B-type) lewis acid behavior. Think of it like a water balloon with many electrons sloshing around from side to side. By following the standard notation, you can avoid ambiguity in parsing and obtain accurate results.\), for example. The cadmium ion is positively charged and posses a large polarizability. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. For example, C2H5OH for ethanol, H2O for water, or Na2SO4 for sodium sulfate. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. They go from 1 to 18 which is the more internationally known numbering system and the official one according to IUPAC. The groups are actually numbered up at the top of the table. A column on the periodic table is known as a group or family. It is important to ensure that the chemical formula is entered correctly using standard notation. The periodic table has all sorts of cool information just based on its layout. Element Tin (Sn), Group 14, Atomic Number 50, p-block, Mass 118.710. The result table includes the element symbol, atomic mass, multiplier (indicating the number of atoms of each element in the formula), and the calculated result. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol). In addition to the molar mass, the calculator also provides a result table with detailed information on the individual elements in the formula. It then calculates the molar mass by adding up the atomic weights of all the elements in the substance. The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents.

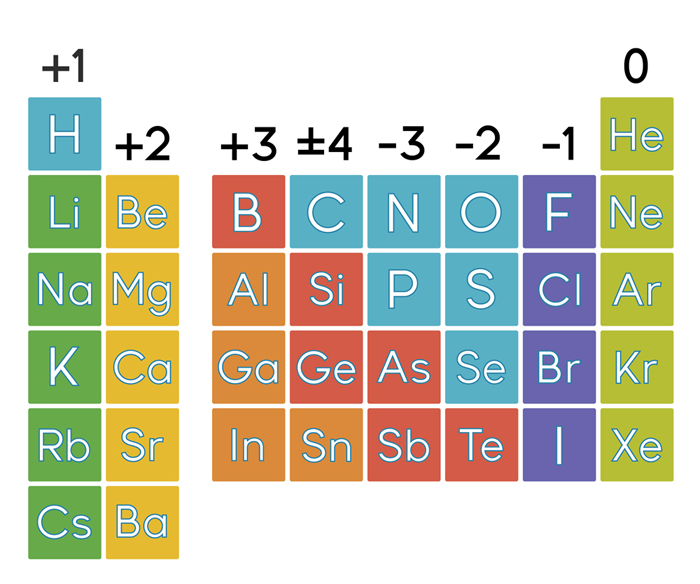
That is, group 1 elements form 1+ ions group 2 elements form 2+ ions, and so on.

Oxygen, we can see from our periodic table of elements, it has a molar mass of 16.00 grams per mole. And then last but not least, we have oxygen here. Hydrogen has a molar mass of 1.008 grams per mole, 008 grams per mole. Moving from the far left to the right on the periodic table, main-group elements tend to form cations with a charge equal to the group number. So carbon has a molar mass of 12.01 grams per mole and now we can think about hydrogen in the same way. Multiply the atomic mass of each element with the number of atoms in the compound then add all the masses. Note the usefulness of the periodic table in predicting likely ion formation and charge (Figure 2.29).

The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs. First, get the atomic mass of Al and Cl from the periodic table. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. To use the calculator, simply input the chemical formula of the substance and click the "Calculate" button. Tin is a chemical element with atomic number 50 which means there are 50 protons in its nucleus. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. The value 55.85 g/mol means that a mole of iron atoms 6.02 x 10 23 (atoms) has a mass of 55.85 grams. In most periodic tables of the elements, the molar mass value is located in the top right hand corner of an elements cell. Element Mercury (Hg), Group 12, Atomic Number 80, d-block, Mass 200.592. This value has been calculated for each element and is usually included in a periodic table. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Created by video journalist Brady Haran working with chemists at The University of Nottingham. The Molar Mass Calculator is a tool used to calculate the molar mass of a substance based on its chemical formula.
